Reducing Submission Time with Veeva Vault RIM
One of the critical needs of the life sciences industry is optimizing the time it takes to submit regulatory documents to health authorities. Using Veeva Vault RIM, life sciences organizations have been able to reduce their regulatory submission (IND, NDA, BLA, etc.) times significantly. The following features of Veeva Vault RIM enable the solution of this problem:
- Comprehensive Ecosystem
- Application Programing Interface (API) Integrations
- Direct Publishing System
- Optimized Submission Process
- Continuous Visibility
- Global Alignment
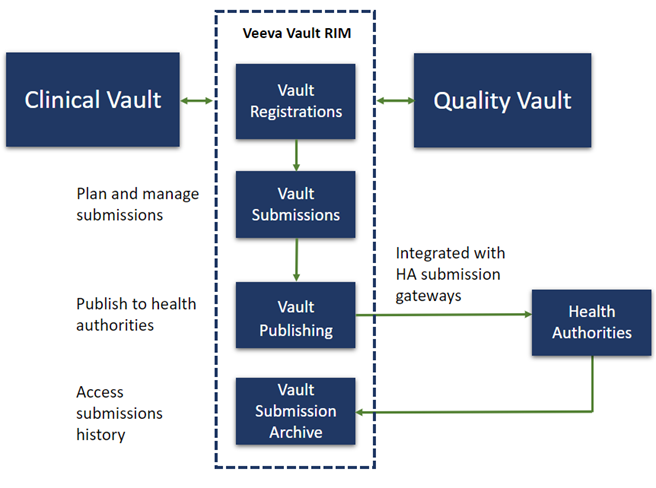
- Comprehensive Ecosystem: Veeva Vault RIM application is a comprehensive ecosystem that enables the transfer of documents and records across different functional areas seamlessly. For instance, records can be transferred from Veeva RIM to the Veeva Clinical and Quality suite bi-directionally. RIM enables the transferring of a product, clinical study, and site records from the Veeva Vault Clinical suite to Veeva Vault RIM. It also automates the creation, versioning, and updating of CrossLink documents and document fields. Veeva enables the synchronous availability of Quality Vault documents in the RIM application. These features reduce the creation and management of duplicate data and enable Vault documents to have a single source of truth within an organization. This reduces the amount of time spent searching for documents and reduces issues with versioning.
- Application Programing Interface (API) Integrations: Veeva Vault RIM’s approach to API integrations enables efficient collaboration among medical, legal, and regulatory (MLR) teams for review, and approval of the various documents. The API integrations provide built-in Digital Asset Management (DAM) to establish a “single source of truth” for documents within Veeva Vaults. The Vault API infrastructure also enables content sharing with internal brand portals as well as controlled content access and distribution. Through the API integrations, real-time progress can be tracked across the content lifecycle. Automatic inspections can be performed between different revisions or new layouts.

- Direct Publishing System: Veeva Vault RIM also reduces the amount of time required to submit documents to health authorities through its direct publishing system. Through integrating with health authority electronic submissions gateways, submissions can be transferred in an easy and intuitive manner. All gateway receipts and responses can be automatically archived.

- Optimized Submission Process: There are several additional key features which can be leveraged to optimize and automate the submissions process. They include:
- Rules-based auto-matching between submission content and submission outline
- Automated background validation and link testing with behind-the-scenes service that identifies problems as submissions are being compiled
- Automated submission-ready rendering for all documents with the correct PDF standards: Navigate web links, cross references, and tables of contents directly from the viewer
- Creation of hyperlinks that are independent of the submission structure so they can be used earlier in the process during document reviews and reused across multiple submissions
- Collaborative authoring with Microsoft Office enables multiple Veeva Vault users to edit an Office document at the same time using Office desktop software, Office Online™, or the Office mobile apps
- Continuous Visibility: Veeva Vault RIM’s continuous visibility feature provides the ability to report on and locate the source of delays in real time. The progress of regulatory submissions and the status of individual documents can be tracked through actionable reports and dashboards which are easy to generate. Reporting and dashboards provide visibility into the source of delays and enables the team to act immediately. Veeva Vault RIM’s reporting features also enables the identification of errors and address broken links to source data as the submission is being built so teams don’t have to go through the re-publishing process multiple times. This is a key feature that significantly reduces the time to submit regulatory documents.
- Global Alignment: Finally, global alignment within Veeva Vault RIM enables faster response time to Health Authority interactions. Global alignment enables the coordination of regulatory efforts across affiliates and partners within one system. The Veeva Vault RIM suite provides the ability to maintain greater control over affiliate submissions and Health Authority interactions, reducing the time required to resolve any potential issues raised by Health Authorities. The risk of missing commitments is greatly reduced using the features such as monitoring of the deadlines and due dates.
In conclusion, these key features of Veeva Vault RIM enable the life sciences industry to save a significant amount of time when submitting regulatory documents to health authorities.
Please note that Veeva Vault is a proprietary trademark of Veeva Systems. Please visit Veeva’s site for more details: www.veeva.com
For more information on the Veeva Vault RIM Suite, please visit Veeva Systems’ website via the following link: Vault RIM Suite | Veeva
You may also be interested in our other blog articles:
- Reduce Time for Study Migration into Veeva Vault – Prana Life Sciences LLC (prana4life.com)
- Using Veeva Vault Training for GxP and Non-GxP Content – Prana Life Sciences LLC (prana4life.com)
- Veeva Vault Implementation Groundwork – Prana Life Sciences LLC (prana4life.com)
For more information, please Contact Us.