An audit trail is a chronological record that provides documentary evidence of the sequence of activities that have affected a specific operation, procedure, event, or device. US regulation 21 CFR Part 11 requires the use of secure, computer-generated, time-stamped audit trails to independently record the date and time of operator entries and actions that create, modify, or delete electronic records.
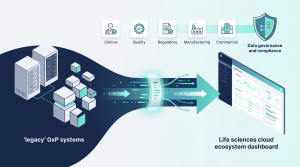
GxP cloud systems produce thousands of audit trail records every day. For compliance, these trails must be periodically reviewed. It is a major challenge to review the vast amount of data that the audit trail records contain. How can you ensure that you have reviewed all the critical information in the audit trail?
We have automated this process to create links from the audit trails to your change requests, change forms, support requests, and other user requests. This enables us to generate the periodic review reports on a daily, monthly, quarterly or on demand basis. The automation is built with a special feature to ensure warning messages are triggered to bring discrepancies to attention.
In conclusion, the automation tool we have created can enable you to ensure that your audit trails are being reviewed in a thorough and consistent manner. Please contact a member of the Prana Life Sciences team for further information.
Also Read,