How Next-Generation Partnerships Are Helping the Industry Navigate the Cloud, Data, and AI Era
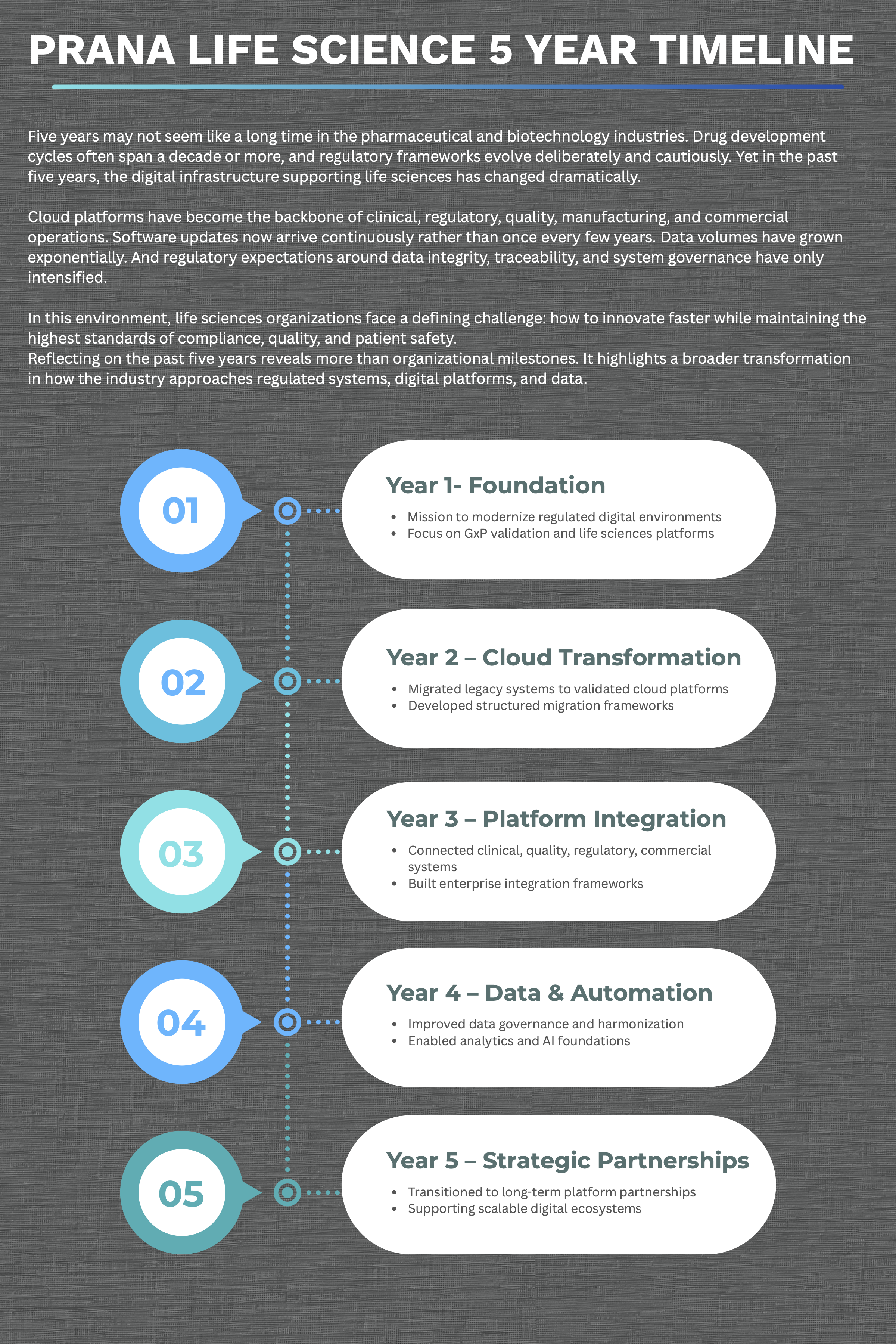
Five years may not seem like a long time in the pharmaceutical and biotechnology industries. Drug development cycles often span a decade or more, and regulatory frameworks evolve deliberately and cautiously. Yet in the past five years, the digital infrastructure supporting life sciences has changed dramatically.
Cloud platforms have become the backbone of clinical, regulatory, quality, manufacturing, and commercial operations. Software updates now arrive continuously rather than once every few years. Data volumes have grown exponentially. And regulatory expectations around data integrity, traceability, and system governance have only intensified.
In this environment, life sciences organizations face a defining challenge: how to innovate faster while maintaining the highest standards of compliance, quality, and patient safety.
Reflecting on the past five years reveals more than organizational milestones. It highlights a broader transformation in how the industry approaches regulated systems, digital platforms, and data.
From Disconnected Projects to Platform-Led Transformation
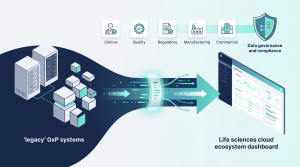
Historically, many life sciences organizations approached technology change as a series of isolated initiatives: a validation effort here, a system migration there, and a new application implementation when legacy systems finally reached end of life.
The result was often a fragmented GxP landscape—multiple systems performing similar functions, disconnected data repositories, and processes that were costly to maintain and difficult to govern.
Over the past several years, a different model has begun to take hold: platform-led digital transformation.
Rather than layering new tools onto aging infrastructure, organizations are increasingly standardizing on modern cloud platforms across clinical, regulatory, quality, and commercial operations. These platforms create integrated ecosystems where data, workflows, and compliance controls operate together rather than in silos.
This shift is fundamentally changing how regulated organizations manage technology. Transformation is no longer about isolated upgrades. It is about building coherent digital foundations that support innovation, governance, and operational scale simultaneously.
Compliance in a World of Continuous Change
While digital platforms have created enormous opportunities, they have also introduced new complexity.
Modern enterprise platforms release enhancements several times per year. Configuration changes can alter workflows quickly. And interconnected systems mean that small updates can ripple across multiple regulated processes.
The traditional model of validation—large, periodic efforts tied to infrequent software releases—no longer aligns with this reality.
Instead, validation has increasingly become a continuous operational discipline embedded within digital environments.
Organizations must now maintain clear traceability between requirements, system configurations, testing activities, and operational evidence across constantly evolving systems. Risk-based validation models, structured impact assessments, and stronger governance frameworks are becoming essential for maintaining inspection readiness while keeping pace with technological change.
The goal is not simply compliance—it is compliance that enables agility rather than constrains it.
Elevating Validation, Migration, and Integration as Strategic Capabilities
In many organizations, validation, migration, and integration were historically viewed as technical necessities—important but largely operational activities.
Over the past several years, however, forward-looking organizations have begun to treat these capabilities as strategic enablers of transformation.
Modern validation approaches must align with cloud release cadences and risk-based methodologies, reducing cycle times while maintaining regulatory confidence. Data migrations from legacy systems must preserve data integrity, traceability, and historical context while enabling organizations to retire aging infrastructure. And system integrations must allow data to move seamlessly across the value chain—from research and development through manufacturing and commercialization.
When these capabilities are executed effectively, organizations gain tangible benefits:
- Reduced dependence on aging, high-risk legacy platforms
- Lower total cost of ownership for regulated systems
- Faster deployment of new capabilities and digital initiatives
In an industry where regulatory missteps can derail programs and erode trust, this combination of rigor and speed represents a meaningful competitive advantage.
Making Life Sciences Data “AI-Ready”
The promise of artificial intelligence in life sciences is widely recognized. AI has the potential to accelerate drug discovery, optimize clinical trials, improve manufacturing quality, and enhance regulatory decision-making.
Yet many organizations face a fundamental obstacle: data readiness.
Advanced analytics and AI systems require structured, high-quality, well-governed data environments. Without these foundations, AI initiatives often remain limited to pilots or isolated proofs of concept.
Over the past five years, leading organizations have focused on harmonizing data across validated systems, improving governance frameworks, and building architectures that make data accessible without undermining regulatory controls.
These efforts enable:
- Standardized data models that support analytics and AI use cases
- Integration patterns that decouple data from legacy applications
- Trusted data pipelines that allow insights to be generated safely within regulated environments
This foundational work is often invisible from the outside, but it is essential. AI transformation in life sciences is only possible when data ecosystems are trustworthy, structured, and compliant.
Redefining Managed Services for Regulated Industries
Another notable shift in the industry has been the evolution of managed services.
In many sectors, managed services have traditionally been associated with commoditized support functions. In life sciences, however, operational support must account for regulatory requirements, audit readiness, and the complexities of highly specialized processes.
A new generation of service models is emerging—ones that combine operational excellence with deep domain expertise in clinical, quality, regulatory, and commercial systems.
These models provide dedicated teams that understand not only the technology platforms but also the regulatory and operational contexts in which they operate. Service delivery becomes aligned with inspection readiness, change control processes, and long-term governance rather than simple system maintenance.
For life sciences organizations, this allows internal teams to focus on scientific innovation and market impact while relying on specialized partners to manage the complexity of regulated digital ecosystems.
Raising Expectations for Industry Partnerships
Taken together, these shifts are redefining what life sciences organizations expect from technology and services partners.
The industry is moving beyond transactional vendor relationships toward long-term strategic collaborations where success is measured by reduced risk, accelerated innovation, and improved outcomes across the development and commercialization lifecycle.
Partners are expected to deliver not only isolated expertise but also integrated capabilities across platforms, data, processes, and compliance frameworks.
Perhaps most importantly, the industry is recognizing that compliance and innovation are not opposing forces. With the right architectures, operating models, and expertise, organizations can achieve both simultaneously.
Looking Ahead: The Next Phase of Digital Life Sciences
The past five years have demonstrated that digital transformation in life sciences is accelerating—and that it must occur within a framework of trust, regulatory rigor, and scientific responsibility.
Looking forward, several trends are likely to shape the next phase of the industry’s evolution:
- Greater adoption of AI-enabled operational and regulatory processes
- Continued migration toward integrated cloud ecosystems
- Increasing emphasis on data governance and traceability
- Stronger alignment between digital infrastructure and scientific innovation
For senior executives across the industry, the question is no longer whether technology will reshape life sciences—it already has.
The real question is how organizations can harness that change safely, efficiently, and strategically.
The past five years have shown that when deep domain expertise meets modern digital platforms, it becomes possible to rethink how regulated systems operate. And in doing so, the industry moves one step closer to its ultimate mission: delivering safe, effective therapies to patients faster and more reliably than ever before.
That is the transformation worth marking—and the one that will define the decade ahead.