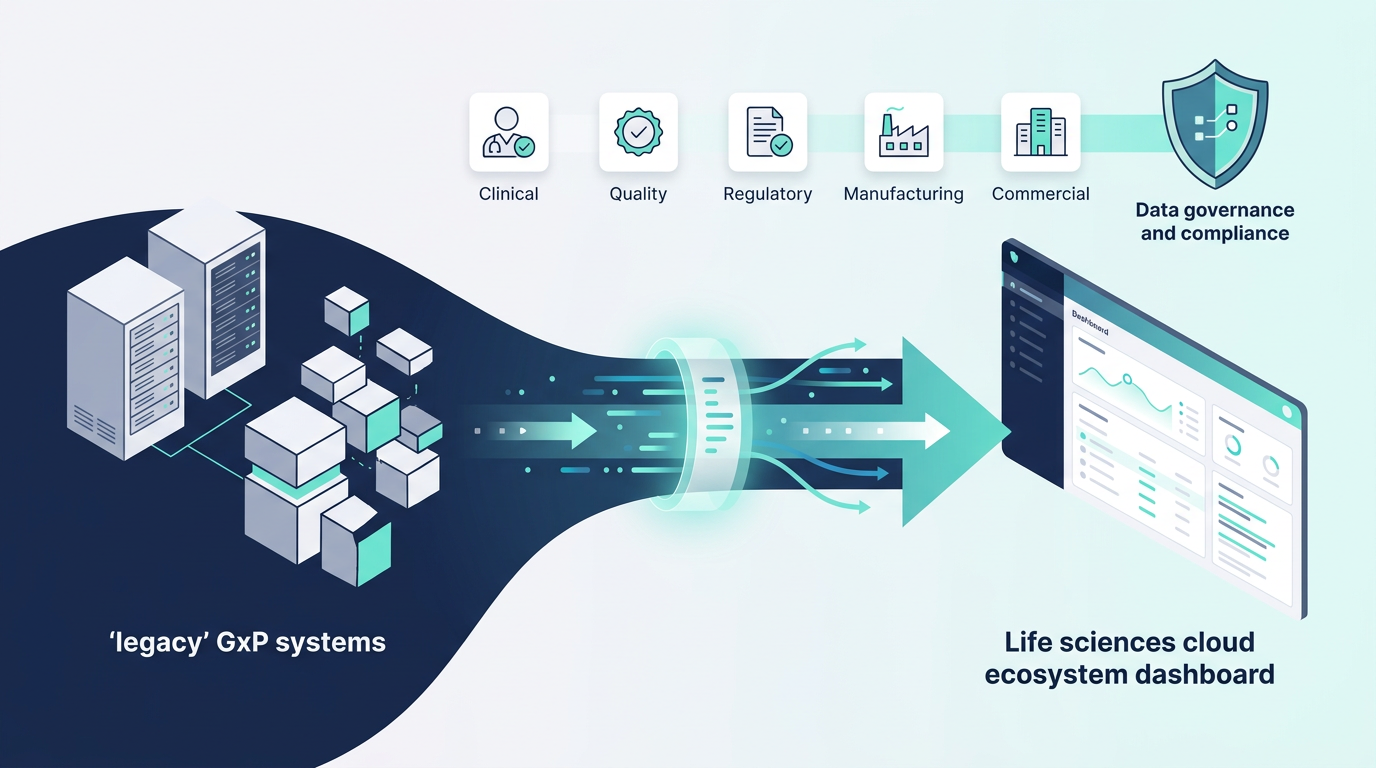
Digital transformation has become a strategic priority for life sciences organizations. Pharmaceutical, biotechnology, and medical device companies are increasingly adopting cloud platforms, advanced analytics, and integrated enterprise systems to support clinical development, quality management, and commercial operations.
However, modernizing regulated technology environments presents a unique challenge. Organizations must transform legacy infrastructure while maintaining strict compliance with regulatory frameworks governing GxP systems, data integrity, and patient safety.
Two activities are central to this transformation: data migration and system integration. Both are technically complex and operationally sensitive in regulated environments. When executed poorly, they can introduce compliance risks, disrupt business processes, and compromise the integrity of regulated data.
When executed well, they enable organizations to modernize safely while strengthening operational control. This article serves as a Practitioners Guide for data migration professionals in the Life Sciences industry.
The Legacy System Challenge in Regulated Environments
Many life sciences organizations operate technology landscapes that have evolved over decades. Legacy applications often support critical regulated processes such as clinical data management, pharmacovigilance, manufacturing quality, and regulatory submissions.
These systems frequently present several limitations:
- Aging infrastructure that is difficult to maintain
- Limited interoperability with modern platforms
- Fragmented data stored across multiple repositories
- Manual processes that increase operational risk
Despite these challenges, legacy systems cannot simply be replaced without careful planning. Regulated systems contain historical GxP data that must remain accurate, traceable, and accessible for regulatory inspections.
As a result, modernization efforts must carefully balance innovation with compliance obligations.
Migrating Legacy GxP Systems Safely
Data migration is often one of the most sensitive phases of a digital transformation initiative. In regulated environments, it involves transferring validated data from legacy systems into modern platforms without compromising data integrity.
A structured migration strategy typically includes several key stages.
Data Assessment and Classification
Before migration begins, organizations must understand the nature of the data stored in legacy systems. This includes identifying:
- GxP-regulated records
- Historical audit trails
- Metadata associated with system transactions
- Relationships between datasets
Not all data requires the same migration approach. Some records must be fully migrated into the new system, while others may be archived in validated repositories for regulatory access.
Data Cleansing and Normalization
Legacy systems often contain inconsistencies such as duplicate records, incomplete fields, or outdated formats. Data cleansing is therefore a critical step prior to migration.
In regulated environments, however, data cleansing must be performed carefully. Changes to historical records must be documented and justified to ensure compliance with data integrity principles.
Migration Verification and Reconciliation
Validation teams must confirm that migrated data accurately reflects the source system. This typically involves reconciliation activities such as:
- Record counts and completeness checks
- Field-level comparisons between systems
- Verification of metadata and audit trails
These activities provide evidence that the migration process preserved data accuracy and traceability.
Validation of the Migration Process
Migration tools, scripts, and workflows themselves must be validated to demonstrate that they perform consistently and reliably.
This validation often includes:
- Test migrations using representative datasets
- Verification of error-handling procedures
- Documentation of repeatable migration protocols
By validating the migration process rather than only the migrated data, organizations strengthen regulatory defensibility.
Integration Across Clinical, Quality, and Commercial Systems
Beyond data migration, modern life sciences environments increasingly rely on integrated system architectures.
Clinical trial platforms, quality management systems, regulatory information systems, manufacturing systems, and commercial applications must often exchange data seamlessly. Integration enables organizations to improve operational visibility and reduce manual data transfer between systems.
However, integration introduces new regulatory considerations.
Managing System Boundaries
In regulated environments, system boundaries must be clearly defined. When systems exchange data, organizations must understand which system is responsible for:
- Data creation
- Data modification
- Data storage
- Regulatory record retention
Clear ownership ensures that compliance responsibilities remain well defined.
Ensuring Data Integrity Across Interfaces
Data transmitted between systems must remain accurate and complete. Integration mechanisms should include controls such as:
- Message validation rules
- Error handling and logging
- Automated reconciliation processes
- Monitoring of interface performance
These controls help ensure that data exchanged across systems maintains integrity throughout its lifecycle.
Validating Interfaces and Integration Workflows
Interfaces between regulated systems must be validated to demonstrate that data flows correctly under expected conditions.
Integration validation typically involves testing scenarios such as:
- Normal data transmission
- Error conditions and exception handling
- Data synchronization across systems
- System recovery following interruptions
By validating these scenarios, organizations can demonstrate that integrated environments operate reliably under real-world conditions.
Data Governance and Traceability
Effective data governance is essential during both migration and integration initiatives.
Regulators expect organizations to maintain clear traceability for regulated data. This means organizations must be able to demonstrate:
- Where data originated
- How data has been transformed or migrated
- Which systems store authoritative records
- Who has accessed or modified the data
Traceability becomes particularly important during regulatory inspections. Inspectors may request evidence showing how historical records were migrated or how data flows between integrated systems.
To support these requirements, organizations should implement governance frameworks that include:
- Data ownership definitions
- Data lifecycle management policies
- Audit trail controls
- Metadata standards for system interoperability
These governance structures help ensure that modernization efforts strengthen rather than weaken compliance posture.
Reducing Operational Disruption During Transformation
Digital transformation initiatives can introduce operational disruption if not managed carefully. Migrating systems and integrating new platforms often requires changes to workflows, training, and organizational processes.
Several strategies can help reduce disruption.
Phased Migration Approaches
Rather than migrating all systems simultaneously, organizations may implement phased migration strategies that prioritize high-value systems while allowing time for validation and stabilization.
Parallel System Operation
In some cases, legacy and modern systems may operate in parallel during transition periods. This approach allows teams to verify system performance before fully decommissioning legacy platforms.
Cross-Functional Governance
Successful transformation initiatives require coordination across IT, quality assurance, regulatory affairs, and operational teams. Governance structures that bring these stakeholders together help ensure that transformation activities align with regulatory expectations.
Change Management and Training
New systems often introduce new workflows. Providing appropriate training and support helps ensure that users adopt new platforms effectively while maintaining compliance practices.
Conclusion
Modernizing technology infrastructure in regulated life sciences environments is both necessary and complex. Legacy systems must be replaced or integrated with modern platforms to support digital transformation, data-driven decision-making, and operational efficiency.
However, modernization cannot occur at the expense of regulatory compliance.
Successful data migration and system integration initiatives depend on structured methodologies that preserve data integrity, traceability, and validation evidence throughout the transformation process.
For senior executives overseeing digital transformation in regulated environments, the strategic objective is clear: modernization initiatives must not only improve technology capabilities but also strengthen the organization’s ability to maintain regulatory control.
When approached with careful governance, rigorous validation practices, and cross-functional collaboration, modernization efforts can enable life sciences organizations to evolve their technology landscapes while maintaining the trust of regulators, partners, and patients.